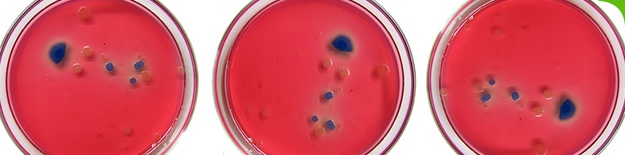
Specified Micro-Organisms
(other names for this test include; TSM, pathogen testing, test for absence of E coli, Salmonella, S aureus, Ps aeruginosa, Bile Tolerant Gram negative Bacteria)
Routine quality control testing is a regulatory requirement. We offer testing to the latest European Pharmacopoeia methods. The EP methods are harmonised with the BP, USP & JP and are therefore interchangeable.
This test determines the presence or absence of certain named micro-organisms. We routinely test for:
- E coli
- Salmonella
- S aureus
- Ps aeruginosa
- Bile Tolerant Gram Negative Bacteria (BTGNB).
This test is commonly used in conjunction with a Total Viable Count (TVC) as the microbial limits test.
Should you need, we are also able to perform testing to your own methods.
For licensed products this test must be validated specifically for your product. See here for more details.
For more information, technical details and sampling advice please see our Technical Information.